Billions of doses of COVID-19 vaccines are being produced and used already – but that is not the end of the story.
According to the World Health Organization's vaccine tracker, there are now a total of 294 vaccines in development, 110 of which are in various stages of clinical development.
So far, the first generation of vaccines have all been delivered by injection, normally in two doses.
Most of the vaccines will be delivered by injection. /Getty Creative via VCG
Most of the vaccines will be delivered by injection. /Getty Creative via VCG
The ones already approved by the WHO are proving remarkably effective at cutting an infected person's chance of being hospitalized, or dying from COVID-19.
They also reduce the chance of becoming infected, or passing on the infection, although studies are continuing to find out by how much they cut transmission.
Vaccines in development by type
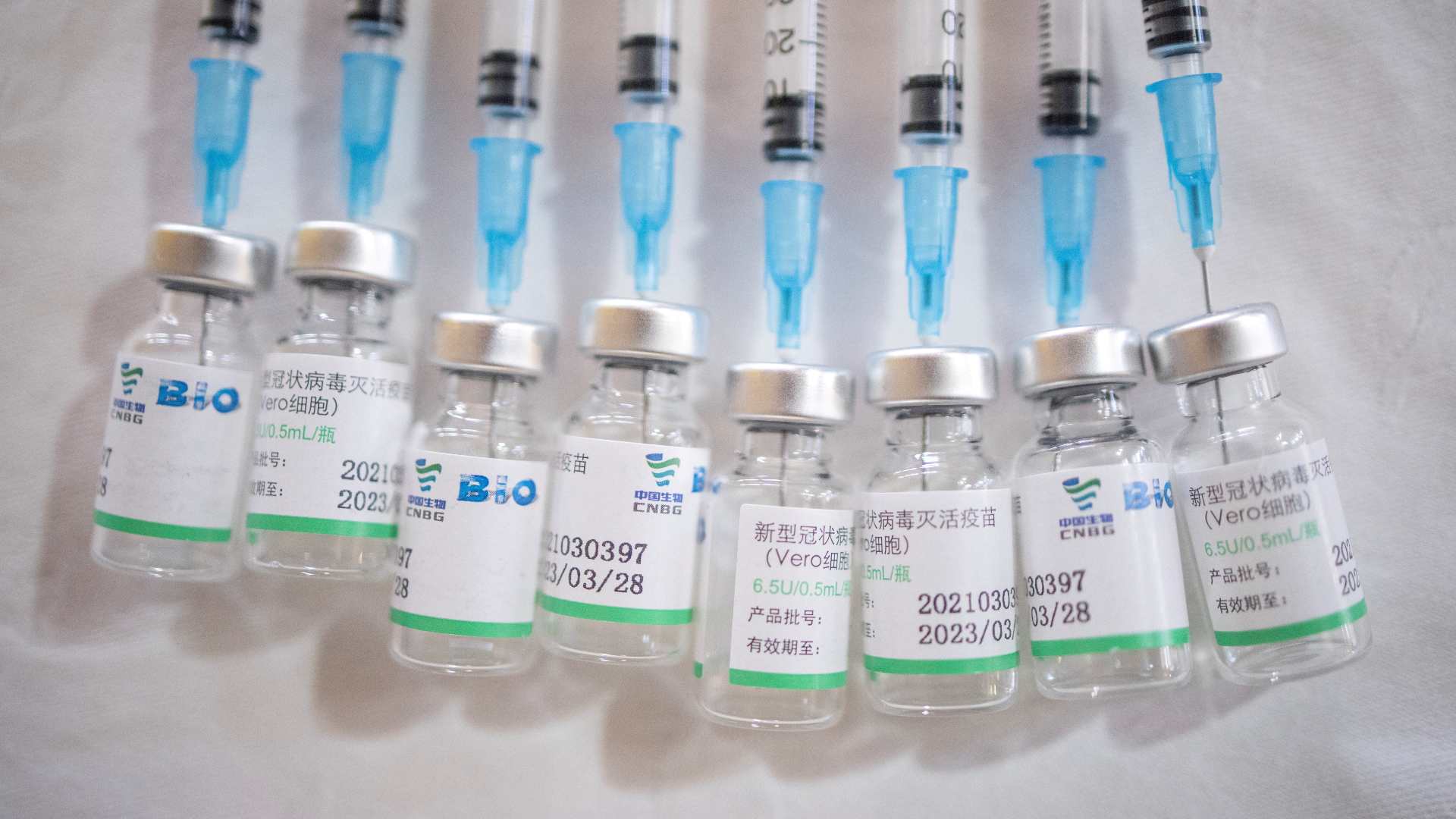
The Sinopharm and Sinovac vaccines have been approved by the WHO. /Reuters
The Sinopharm and Sinovac vaccines have been approved by the WHO. /Reuters
Viral vector (non-replicating) - There are 17 of this type of vaccine in clinical development and 20 in pre-clinical development, according to the WHO. This is the same type as the current AstraZeneca vaccine.
RNA - There are 18 vaccines in clinical development and 23 in pre-clinical development. This is the same type as the current Pfizer-BioNTech vaccine, which is a messenger RNA, or mRNA, jab.
Protein subunit - There are 37 vaccines in clinical development, which is more than a third of the total. There are also 69 in pre-clinical development. This is the type developed by Novavax and Sanofi-GSK, which are currently under review by European regulators for approval.
Inactivated virus - There are 16 vaccines in clinical development and eight in pre-clinical development. This is the type used by Sinovac and Sinopharm.
DNA - There are 10 vaccines in clinical development and 16 in pre-clinical development. According to the Times of India, Zydus Cadila has applied for emergency use authorization, after successful trials.
READ MORE: What are the differences between the current COVID-19 vaccines?
One, two or three doses: How they compare
Two shot doses - This category includes two-thirds of the vaccines in clinical development. Of the 72 double-dose vaccines, a four-week gap between doses is favored for 38, a three-week gap by 28 and a two-week gap between doses for six of them.
Single dose - There are 16 vaccines in this category, which is 15 percent of the total.
Triple dose - There is one vaccine in clinical development, from Zydus Cadila, with a planned three-dose strategy, with doses four weeks apart.
Others - There are 21 of the vaccines in clinical development that are not included in these figures – either because the information was unavailable or because the doses are to be decided after the clinical development process.
If all goes well, vaccines may not always have to involved injections. /Getty Creative via VCG
If all goes well, vaccines may not always have to involved injections. /Getty Creative via VCG
Eighty five – the vast majority – of the vaccines in clinical development will be delivered to people by injection. This is the way the currently approved COVID-19 vaccines are delivered.
There are eight vaccines in clinical development that will be delivered nasally. This is a delivery system that has been widely used for things such as flu vaccines for young children.
There are three vaccines in clinical development that would be taken orally. This method has been used for vaccines including polio.
Other – There are 14 of the vaccines in clinical development for which the World Health Organization says the method of delivery is yet to be announced.

